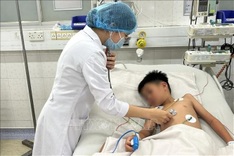
Each volunteer will receive two doses of either the vaccine or the placebo AIPO4, with an interval of 28 days between each dose. The volunteers will then be monitored for 12 months after receiving their first dose.
Volunteers who were given their initial shots on the morning of February 26 will then receive their second doses in late March.
Lieutenant General Do Quyet, director of the Military Medical University, said since the trial involves the participation of numerous volunteers suffering from underlying health conditions, competent authorities have prepared for a range of scenarios to ensure the safety of all volunteers.
Results of the trial are set to be announced in May before preparing for the third stage of trials, during which only one single shot of the vaccine will be administered to between 10,000 and 15,000 people from both domestic and foreign pandemic-hit regions, Lieut. Gen. Quyet added.
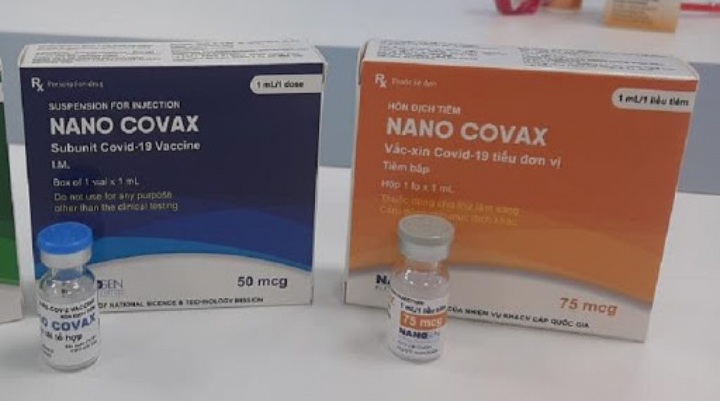
Originally developed by the Nanogen Pharmaceutical Biotechnology JSC and the Hanoi-based Military Medical University, Nano Covax represents the nation’s first COVID-19 vaccine which has successfully reached the human trial stage.
The initial stage of trials of the Nano Covax vaccine indicates that it is likely to be effective against the B117 variant from the UK.
The first domestically-produced vaccine Nanocovax, studied by the Nanogen Pharmaceutical Biotechnology JSC, has now entered the second phase of clinical trials. By the end of March 4, the phase two study had reached 65%. Elsewhere, Vabiotech is the third research unit of the COVID-19 vaccine, although it has not submitted a clinical trial application at this point.