If the human trials are successful, the firm will manufacture some 2 million doses on a trial basis in the form of injections, nasal spray, and eye drops, to meet the needs of different age groups.
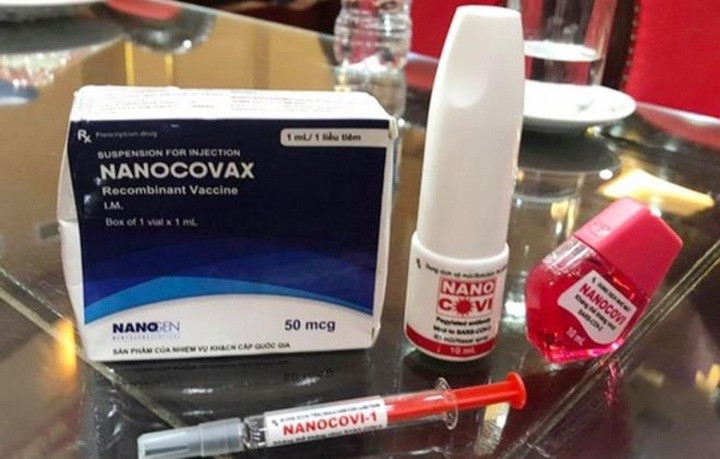
If the human trials are successful, the firm will manufacture some 2 million doses on a trial basis in the form of injections, nasal spray, and eye drops, to meet the needs of different age groups.
Once developed, individuals suffering from underlying diseases and children under the age of 12 will be able to make use of the vaccine.
At present, the company is recruiting volunteers for first human clinical trials of its Nano Covax vaccine, with the first stage scheduled to start on December 17.
This recombinant vaccine has been already been tested on mice, hamsters, and monkeys, so far showing a good immune response. As a result, it has been approved by the Ministry of Health to progress to human clinical trials.
If the human trials prove to be both safe and effective, the vaccine will be rolled out commercially in the middle of 2021.